
In a compound, there is a variable composition of the elements. True or false? Explain your answer based on your observations. When elements combine to form compounds, the physical properties of the compound are the same as those of the elements. Formula of compound Example: CuSO4.5H20 the Physical properties Physical Properties of each element in the of the compound compound Deep blue Copper-a yellow-brown, shiny solid crystalline Sulfur-a yellow, powdery solid solid Oxygen-a colorless gas Hydrogen-a colorless gas 1. Sulfur is a nonmetal with a distinct odor and is used in the production of sulfuric acid, fertilizers, and rubber products. Hydrogen, for example, is the lightest and most abundant element in the universe, and it is commonly used as fuel for rockets and as a reactant in chemical reactions. Observe the display of elements and record the physical properties of each element in the compound. Some reactive nonmetals have unique and notable properties. Give the full name of each element in the compound. 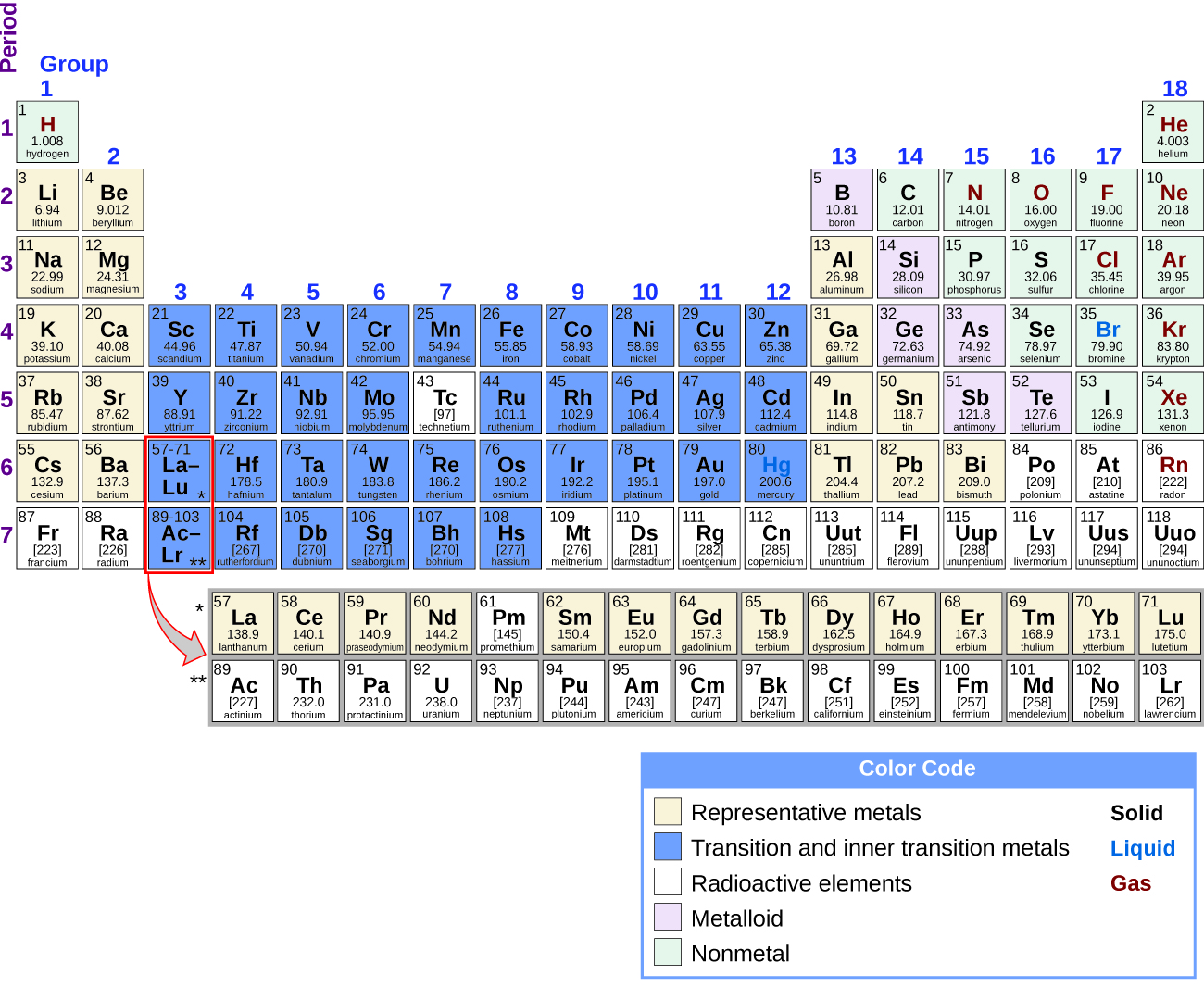
Describe the physical properties of each compound including color, luster, and phase (solid, liquid or gas). Record the chemical formula given for each compound. PHYSICAL PROPERTIES OF ELEMENTS AND COMPOUNDS Observe the display of chemicals. A jagged black line in a periodic table (see figure below) along the right side of the table separates the metals from the nonmetals. This type of classification is based on the shared physical and chemical properties. (b) Classify the following into metals, non-metals and metalloids: Silicon, mercury, Diamond, Sulphur, Iodine, Germanium, Sodium, carbon, magnesium, Copper. Although rarely used in pure form, manganese is essential to steelmaking. They are located on the right side of the periodic table. If they are solid, they tend to be brittle. Non-metals can be solids, liquids, or gases. They are located on the left side of the periodic table. Transcribed image text: Identify each element as a metal, non-metal, or metalloid. It was recognized as an element in 1774 by the Swedish chemist Carl Wilhelm Scheele. Metals tend to be shiny, hard, malleable and good conductors of electricity. Element Symbol Atomic Number Color Luster Metal or Nonmetal aluminum carbon copper iron nitrogen magnesium manganese nickel oxygen phosphorus potassium silicon gold silver tin zinc cobalt lead chlorine Osmium cesium antimonyĢ6 C. Metals and non-metals are generally heard of but here we will also study the intermediate type of elements called metalloids. Manganese, chemical element that is a silvery white, hard, brittle metal of Group 7 in the periodic table. 
25 B PHYSICAL PROPERTIES OF THE ELEMENTS Observe the elements in the Periodic Table display and fill in the following table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
